Case studies
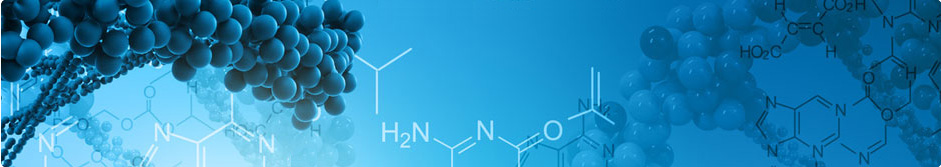
Abacavir
A potent anti-HIV drug received FDA approval in 1998. Abacavir is a nucleoside reverse transcriptase inhibitor (NRTI), available in tablet and oral solution form. It works by slowing the spread of HIV in the body. Abacavir does not cure HIV infection and may not prevent you from developing HIV-related illnesses. Abacavir does not prevent you from spreading HIV infection to other people.
Download PDF
| Contact us at for a copy of the full report, or to request a complimentary Real Time Intelligence report on one of your drugs. |
Avandia:
For
treating type 2 diabetis mellitus
 |
Approval: 1999
Active Ingredient: Rosiglitazone maleate
Number of label changes (1 yr.): none
Number of label changes (Total): 16
Black box Warning: Yes
On Active FDA Safety Alert: Yes
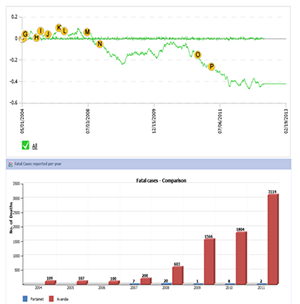
Rosiglitazone is an anti-diabetic drug approved for improving glycemic control in type 2 diabetes mellitus patients. The FDA has issued a black box warning for increased risk of congestive heart failure and myocardial infarction associated with the usage of Avandia. |
It is not recommended for patients with symptomatic heart failure. It is advised that the diabetic patients using Avandia be monitored for rapid weight gain, shortness of breath, and/or edema. The benefit/risk profile of the drug (top left) agrees with the increasing number of fatal cases reported for Avandia (bottom left, red; FDA AERS database). Volume of such cases due to another anti-diabetic drug Fortamet (metformin) with black box warning for lactic acidosis, has been used to indicate the magnitude of adverse effects reported among Avandia users.
Ziagen: Antiviral medication to treat human immunodeficiency virus
 |
Approval: December 1998
Active Ingredient: Abacavir sulfate
Number of label changes (1 yr.): 2
Number of label changes (Total): 15
Black box Warning: Yes
On Active FDA Safety Alert: Yes
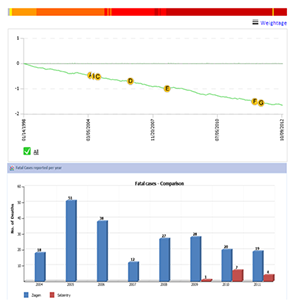
Ziagen, a nucleoside reverse transcriptase inhibitor, was approved for the treatment of HIV-1 infection. The drug has a black box warning for multi-organ hypersensitivity reactions, severe hepatomegaly, and lactic acidosis. |
Hypersensitivity strongly correlates with a histocompatibility antigen HLA-B*5701. The black box warning recommends that all patients must be screened for HLA-B*5701 before starting treatment with abacavir or abacavir-containing combination drugs. The RTI, which indicates a benefit/risk profile of a drug (top left), shows a trend of increasing risk with using abacavir sulfate. Selzentry, another anti-HIV-1 drug, also has a black box warning for hepatotoxicity. However, as per the FDA AERS database, the number of adverse reports and cases of fatality due to Selzentry is several fold less than Ziagen (bottom left). |
|
 |